braz
j
infect
dis
2
0
2
0;2
4(3):247–249
www.elsevier.com/locate/bjid
The
Brazilian
Journal
of
INFECTIOUS
DISEASES
Case
report
An
infant
with
a
mild
SARS-CoV-2
infection
detected
only
by
anal
swabs:
a
case
report
Juan
Li
a
,
Jing
Feng
a
,
Tian-hu
Liu
b
,
Feng-cheng
Xu
b
,
Guo-qiang
Song
c
,∗
a
Pidu
District
People’s
Hospital,
Department
of
Infectious
Diseases,
Chengdu,
Sichuan
Province,
China
b
Pidu
District
People’s
Hospital,
Department
of
Cardiology,
Chengdu,
Deyuan
Chengdu,
Sichuan
Province,
China
c
Changxing
County
Hospital,
of
Traditional
Chinese
Medicine,
Department
of
Respiratory
Medicine,
Huzhou,
Zhejiang
Province,
China
a
r
t
i
c
l
e
i
n
f
o
Article
history:
Received
20
March
2020
Accepted
20
April
2020
Available
online
6
May
2020
Keywords:
Severe
acute
respiratory
syndrome
coronavirus
2
Baby
Anal
swab
a
b
s
t
r
a
c
t
Severe
acute
respiratory
syndrome
coronavirus
2
(SARS-CoV-2)
emerged
in
Wuhan,
China
and
has
spread
rapidly
worldwide.
We
present
a
mild
SARS-CoV-2
infection
in
a
baby
with
non-productive
cough
and
normal
chest
computed
tomography,
in
whom
only
anal
swabs
tested
positive
by
real-time
PCR
testing
for
SARS-CoV-2.
She
was
given
atomization
inhalation
therapy
with
recombinant
human
interferon
alfa-1b
for
10
days.
Her
anal
swabs
remained
positive
for
eight
days,
whereas
her
throat
swabs
were
persistently
negative
by
real-time
PCR
testing.
Mild
and
asymptomatic
cases,
especially
in
children,
might
present
with
PCR
negative
pharyngeal/nasal
swabs
and
PCR
positive
anal
swabs.
Those
patients
are
potential
sources
of
infection
via
fecal–oral
transmission
for
COVID-19.
©
2020
Sociedade
Brasileira
de
Infectologia.
Published
by
Elsevier
Espa
˜
na,
S.L.U.
This
is
an
open
access
article
under
the
CC
BY-NC-ND
license
(http://creativecommons.org/
licenses/by-nc-nd/4.0/
).
Introduction
Severe
acute
respiratory
syndrome
coronavirus
2
(SARS-CoV-
2)
emerged
in
Wuhan
and
has
spread
rapidly
in
China,
South
Korea,
and
worldwide.
Since
December
2019,
more
than
120,000
people
have
been
infected.
1–3
The
SARS-CoV-2
genome
has
a
nucleotide
identity
of
89%
with
bat
SARS-like-
CoVZXC21
and
82%
with
human
SARS-CoV.
4
The
most
common
symptoms
of
infection
are
fever
(43.8%
on
admission
and
88.7%
during
hospitalization)
and
cough
(67.8%).
Diarrhea
is
uncommon
(3.8%).
The
median
incubation
period
is
four
days
(interquartile
range
2–7).
On
admission,
∗
Corresponding
author.
E-mail
address:
(G.
Song).
ground-glass
opacities
are
the
most
common
radiological
finding
on
chest
computed
tomography
(CT)
(56.4%).
No
radio-
graphic
or
CT
abnormality
was
found
in
157
of
877
patients
(17.9%)
with
non-severe
disease
or
in
5
of
173
patients
(2.9%)
with
severe
disease.
Lymphocytopenia
was
present
in
83.2%
of
the
patients
on
admission.
5
The
diagnosis
is
confirmed
by
testing
pharyngeal
or
nasal
swabs
for
viral
nucleic
acids.
In
a
Chinese
Center
for
Disease
Control
and
Prevention
(CDC)
report,
the
overall
case-fatality
rate
was
2.3%,
with
1023
deaths
among
44,672
confirmed
cases.
6
The
pandemic
has
caused
many
social
public
health
problems,
leading
to
eco-
nomic
recession
and
panic.
7
This
is
a
global
health
problem
and
not
just
a
problem
in
China.
https://doi.org/10.1016/j.bjid.2020.04.009
1413-8670/©
2020
Sociedade
Brasileira
de
Infectologia.
Published
by
Elsevier
Espa
˜
na,
S.L.U.
This
is
an
open
access
article
under
the
CC
BY-NC-ND
license
(http://creativecommons.org/licenses/by-nc-nd/4.0/).
248
b
r
a
z
j
i
n
f
e
c
t
d
i
s
.
2
0
2
0;2
4(3):247–249
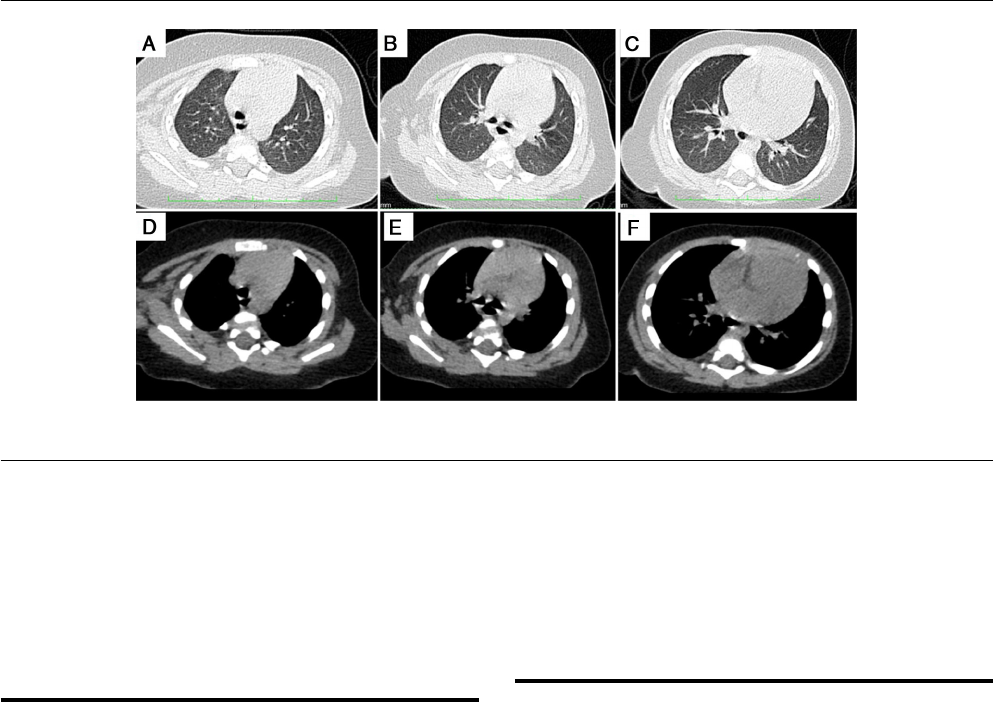
Fig.
1
–
Normal
chest
CT
findings
at
different
sections:
A
and
D,
B
and
E,
and
C
and
F
represent
the
same
levels.
While
patients
with
positive
pharyngeal
or
nasal
swabs
have
received
much
attention,
asymptomatic
or
mildly
ill
patients
with
positive
anal
swabs
have
not.
We
know
little
about
these
patients,
who
may
be
shadows
in
the
sun.
How-
ever,
they
are
potential
sources
of
infection
via
fecal–oral
transmission.
Here,
we
present
an
asymptomatic
baby
in
whom
anal
swabs
were
positive
while
pharyngeal
swabs
were
persistently
negative
by
real-time
PCR
testing.
Case
report
An
8-month-8-day-old
girl
was
hospitalized
with
one
day
his-
tory
of
cough
and
runny
nose
on
Feb
25,
2020.
The
patient
was
afebrile
with
no
shortness
of
breath,
clubbing,
cyanosis,
or
abdominal
distension.
The
patient
was
given
oral
ambroxol
hydrochloride
solution
15
mg,
three
times
per
day,
with
no
improvement.
The
patient’s
mother,
diagnosed
with
SARS-
CoV-2
2
days
earlier,
had
positive
pharyngeal
swab
by
real-time
PCR
testing
and
chest
CT
showing
ground-glass
opacities.
On
physical
examination,
the
patient
had
a
body
tem-
perature
of
38
◦
C,
pulse
of
116
beats/min,
respiratory
rate
of
30
breaths/min,
and
oxygen
saturation
in
room
air
of
99%.
She
was
conscious
and
appeared
acutely
ill.
There
were
no
fluctua-
tions
in
the
nasal
wings
but
some
secretion
in
the
nasal
cavity,
with
redness,
congestion,
and
a
normal
voice
was
observed.
The
breath
sounds
were
normal
in
both
lungs,
with
no
dry
or
wet
rales.
Heart
auscultation
was
normal.
The
abdomen
was
soft,
and
bowel
sounds
were
normal.
Laboratory
tests
were
negative
for
influenza
A
and
B
viral
antigens.
The
leukocyte
count
was
11.91
×
10
9
/L,
with
a
neu-
trophil
count
of
2.91
×
10
9
/L
(24.4%)
and
lymphocyte
count
of
7.87
×
10
9
/L
(66.1%).
The
C-reactive
protein
level
was
3.49
mg/L.
The
prothrombin
time
and
d-dimer
level
were
normal,
as
well
as
alanine
transaminase,
alanine
transaminase,
urea
nitro-
gen,
and
creatinine
levels.
Chest
CT
results
had
no
abnormal
findings
(Fig.
1).
Recombinant
human
interferon
alfa-1b
via
atomization
inhalation
(20
g,
twice/day)
was
initiated.
Anal
swabs
were
positive
on
February
27
and
March
4.
The
symptoms
improved
on
March
7,
and
anal
swabs
were
negative
on
March
9
and
10.
The
patient
was
discharged
on
March
11.
Throat
swabs
were
persistently
negative
throughout
the
hospital
stay.
Real-time
PCR
for
COVID-19
on
pharyngeal
and
anal
swabs
were
performed
by
the
Pengzhou
(Sichuan
Province,
China)
CDC.
The
Chengdu
CDC
repeated
the
PCR
testing
and
obtained
the
same
results.
Discussion
Diagnosis
of
SARS-CoV-2
depends
on
imaging,
epidemiolog-
ical
history,
and
nucleic
acid
testing.
During
the
early
stage
of
the
pandemic,
most
patients
were
in
China,
especially
in
Wuhan,
Hubei
Province.
8
As
the
number
of
patients
grew
geo-
metrically,
the
Chinese
government
locked
down
Wuhan
and
effectively
isolated
infected
people
from
non-infected
people.
In
early
March
2020,
the
disease
was
effectively
controlled
in
China.
9,10
However,
many
confirmed
and
suspected
cases
have
appeared
in
other
regions
and
countries.
11
It
has
become
a
global
pandemic,
and
its
rapid
spread
and
high
lethality
require
special
attention.
12
Although
the
infection
and
mortality
rates
are
lower
in
infants
and
young
children
than
in
adults,
8
young
patients
cannot
communicate
effectively,
which
limits
the
ability
to
obtain
their
medical
history.
Our
patient
developed
a
sudden
onset
cough,
and
her
mother
was
confirmed
to
be
infected.
Although
the
baby’s
pharyngeal
swabs
and
chest
CT
were
neg-
ative,
her
anal
swabs
remained
positive
for
eight
days.
Not
enough
attention
is
given
to
asymptomatic
or
mildly
infected
patients
with
positive
anal
swabs.
Here,
we
present
a
mildly
ill
baby
whose
anal
swabs
were
real-time
PCR
positive
for
SARS-CoV-2,
while
many
pharyngeal
swabs
were
nega-
tive.
SARS-CoV-2
was
stable
under
the
conditions
tested.
13
The
main
host
receptor
is
angiotensin
converting
enzyme
2,
which
is
located
on
gastrointestinal
epithelial
cells,
and
the
feces
of
20%
of
SARS-CoV-2
patients
remain
positive
for
viral
RNA
after
negative
conversion
of
viral
RNA
in
the
respiratory
tract.
14
Wang
et
al.
detected
live
SARS-CoV-2
in
stool
samples
from
two
confirmed
patients.
15
Unfortunately,
our
laboratory

b
r
a
z
j
i
n
f
e
c
t
d
i
s
.
2
0
2
0;2
4(3):247–249
249
lacked
the
capacity
to
determine
whether
the
stool
virus
was
still
active.
Physicians
should
be
aware
that
asymptomatic
or
mildly
ill
children
with
history
of
exposure
and
negative
pha-
ryngeal/nasal
swabs
and
positive
anal
swabs
are
potential
sources
of
infection
via
fecal–oral
transmission
for
COVID-
19.
In
addition
to
nasal/pharyngeal
swabs,
we
believe
that
children
should
be
tested
for
SARS-CoV-2
using
anal
swabs.
Conclusion
Infants
with
a
history
of
SARS-CoV-2
exposure
and
mild
symp-
toms
should
be
tested
using
anal
swabs.
Ethical
approval
The
study
was
approved
by
Pidu
District
People’s
Hospital
(Batch
2020-03-18-001).
Funding
No
founding.
Conflicts
of
interest
The
authors
declare
no
conflicts
of
interest.
Informed
consent
This
study
was
a
retrospective
observational
nature
study,
patient
identity
remained
anonymous,
and
have
no
invasive
procedure,
so
the
ethics
committee
waived
informed
consent.
r
e
f
e
r
e
n
c
e
s
1.
Wu
Z,
McGoogan
JM.
Characteristics
of
and
important
lessons
from
the
coronavirus
disease
2019
(COVID-19)
outbreak
in
China:
Summary
of
a
report
of
72,314
cases
from
the
Chinese
Center
for
Disease
Control
and
Prevention.
JAMA.
2020,
http://dx.doi.org/10.1001/jama.2020.2648.
2.
Ki
M.
Epidemiologic
characteristics
of
early
cases
with
2019
novel
coronavirus
(2019-nCoV)
disease
in
Republic
of
Korea.
Epidemiol
Health.
2020,
http://dx.doi.org/10.4178/epih.e2020007.
3.
Patel
A,
Jernigan
DB.
Initial
public
health
response
and
interim
clinical
guidance
for
the
2019
novel
coronavirus
outbreak
–
United
States,
December
31,
2019
–
February
4,
2020.
MMWR
Morb
Mortal
Wkly
Rep.
2020;69:140–6.
4.
Chan
JFW,
Kok
KH,
Zhu
Z,
et
al.
Genomic
characterization
of
the
2019
novel
human-pathogenic
coronavirus
isolated
from
a
patient
with
atypical
pneumonia
after
visiting
Wuhan.
Emerg
Microbes
Infect.
2020;9:221–36.
5.
Chan
JF-W,
Kok
K-H,
Zhu
Z,
et
al.
Genomic
characterization
of
the
2019
novel
human-pathogenic
coronavirus
isolated
from
a
patient
with
atypical
pneumonia
after
visiting
Wuhan.
Emerg
Microbes
Infect.
2020;9:221–36.
6.
Novel
Coronavirus
Pneumonia
Emergency
Response
Epidemiology
Team.
[The
epidemiological
characteristics
of
an
outbreak
of
2019
novel
coronavirus
diseases
(COVID-19)
in
China].
Zhonghua
Liu
Xing
Bing
Xue
Za
Zhi.
2020,
http://dx.doi.org/10.3760/cma.j.issn.0254-6450.2020.02.003.
7.
Johnson
HC,
Gossner
CM,
Colzani
E,
et
al.
Potential
scenarios
for
the
progression
of
a
COVID-19
epidemic
in
the
European
Union
and
the
European
Economic
Area,
March
2020.
Eurosurveillance.
2020;25:1–5.
8.
Wang
Z,
Yang
B,
Li
Q,
Wen
L,
Zhang
R.
Clinical
features
of
69
cases
with
coronavirus
disease
2019
in
Wuhan,
China.
Clin
Infect
Dis.
2020,
http://dx.doi.org/10.1093/cid/ciaa272.
9.
Lin
Q,
Zhao
S,
Gao
D,
et
al.
A
conceptual
model
for
the
coronavirus
disease
2019
(COVID-19)
outbreak
in
Wuhan,
China
with
individual
reaction
and
governmental
action.
Int
J
Infect
Dis.
2020;93:211–6.
10.
Lau
H,
Khosrawipour
V,
Kocbach
P,
et
al.
The
positive
impact
of
lockdown
in
Wuhan
on
containing
the
COVID-19
outbreak
in
China.
J
Travel
Med.
2020;1,
http://dx.doi.org/10.1093/jtm/taaa037.
11.
Hunter
P.
The
spread
of
the
COVID-19
coronavirus.
EMBO
Rep.
2020;(March):e50334.
12.
Remuzzi
A,
Remuzzi
G.
COVID-19
and
Italy:
what
next?
Lancet.
2020;2:10–3.
13.
van
Doremalen
N,
Bushmaker
T,
Morris
DH,
et
al.
Aerosol
and
surface
stability
of
SARS-CoV-2
as
compared
with
SARS-CoV-1.
N
Engl
J
Med.
2020;(March),
http://dx.doi.org/10.1056/NEJMc2004973.
14.
Xiao
F,
Tang
M,
Zheng
X,
Liu
Y,
Li
X,
Shan
H.
Evidence
for
gastrointestinal
infection
of
SARS-CoV-2.
Gastroenterology.
2020;(March),
http://dx.doi.org/10.1053/j.gastro.2020.02.055.
pii:S0016-5085(20)30282-1.
15.
Wang
W,
Xu
Y,
Gao
R,
et
al.
Detection
of
SARS-CoV-2
in
different
types
of
clinical
specimens.
JAMA.
2020:3–4,
http://dx.doi.org/10.1001/jama.2020.3786.
